Log In
Log InSign up
Registration is for professionals that work with containment and Isolation Technologies and grants full access to our product data sheets and catalogues
Sign up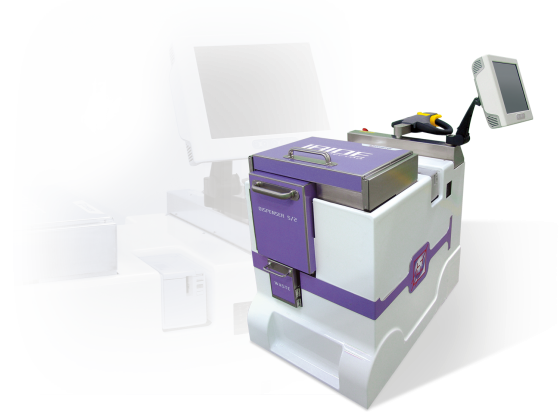
IRIDE can inject patients with a FDG single-dose starting from a multi-dose vial. It is compact, easy to move (it has a motorised main wheel) and is battery operated.
The radioactive product is completely contained in a sterile/disposable kit; after each patient injection only the terminal part must be changed.
FDG total activity is measured by an ionization chamber.
The FDG multi-dose vial is directly loaded into the machine with its own shipping container. IRIDE is designed for use with most commercially available containers. Max FDG multi-dose vial activity is 700 mCi.
IRIDE has no limitation on FDG initial volume or concentration.
The entire content of the multi-dose vial is transferred into a 250 ml bulk container; the multi-dose vial is then completely rinsed with saline solution and emptied.
The FDG activity and volume (by weight) are measured, so activity concentration is accurately calculated (precision better than 2%).
Optimal bulk concentration is lower than 20 mCi/ml. In case of higher concentration, a dilution can be performed for better precision.
IRIDE can precisely dilute the FDG using a motorised syringe. After dilution, the bulk is accurately homogenised and all kit lines are primed. Remaining activity and concentration values are continuously displayed on the 15” command touch screen. The products are transferred via electrically operated three-way valves to the different kit end points.
The three-way valves operate with feedback signals.
The kit has two motorised syringes: one 10 ml is used for dilution and mixing, while a 3 ml is used for injection.
IRIDE may be connected to the Hospital network to acquire patient data. After each injection, a report is printed.
Log in to your Comecer Account to download data sheets
Log In Lost PasswordNews and invitations to events and fairs directly in your mailbox
Subscribe
Technical

Statistical

Targeting